Forensic-Level Analysis of Foreign Matter and Impurities
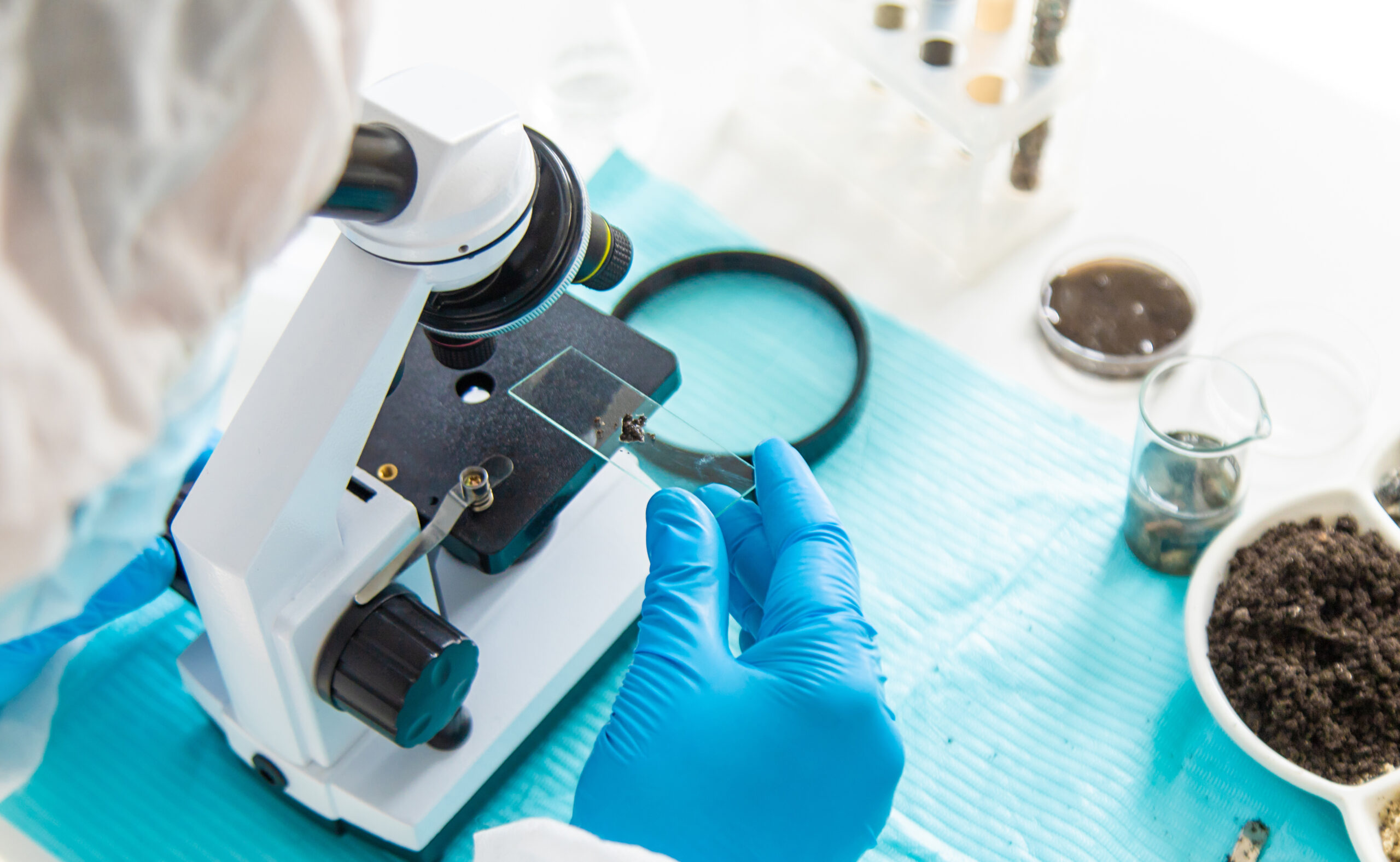
Contaminant Identification is essential when unknown particles, residues, or discolorations appear in raw materials, intermediates, or finished products. Whether metallic, organic, or particulate in nature, these contaminants can compromise product safety, reliability, and brand trust.
BQCIS employs a multi-instrumental analytical approach combining advanced microscopy, spectroscopy, and chromatography to identify the chemical and physical origin of contamination. Our methods provide root-cause clarity—supporting product recalls, supplier qualification, and process improvement.
Results are traceable, reproducible, and compliant with international quality frameworks such as ISO/IEC 17025, ASTM E1613, and ICH Q6A.
Key Activities in Contaminant Identification
Key Benefits of Contaminant Identification

Root Cause Resolution
Provides forensic evidence linking contamination to its origin within the production or supply process.
Quality Preservation
Prevents reoccurrence through corrective and preventive actions, safeguarding brand integrity.

Regulatory Assurance
Supports documentation and verification for FDA, EMA, and ISO 9001 compliance investigations.

Process Optimization
Enhances manufacturing reliability through data-driven contamination control strategies.
Success Story
Contamination Source Analysis in Beverage Production
A global beverage manufacturer discovered fine particulate contamination in bottling lines, affecting clarity and shelf stability.
BQCIS used SEM-EDX and FTIR analysis to identify the contaminant as a mixture of glass and polymer particles originating from cap-molding residue.
Supplier tooling adjustments eliminated the defect, restoring product quality and preventing losses exceeding USD 1.2M per production run.
